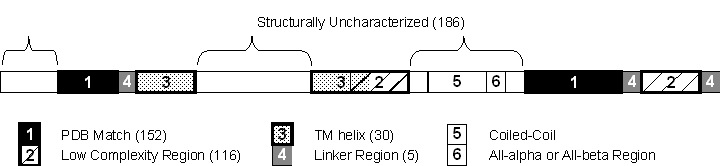
|
Abbrev. |
Num |
Full Mask Name |
Sequence file IN |
Sequence file OUT
(MBY = masked by) |
|
pdb |
1 |
minscop soluble matches |
seq |
seq MBY pdb |
|
pdn |
1 |
minscop soluble matches no overlap (best choice for PDB matches) |
seq |
seq MBY pdb |
|
pdo |
1 |
minscop soluble matches overlap |
seq |
seq MBY pdo |
|
lcl |
2 |
low complexity long |
seq |
seq MBY lcl |
|
lcl |
2 |
low complexity long |
seq MBY pdb |
seq MBY pdb MBY lcl |
|
tms |
3 |
tm segs |
seq |
seq MBY tms |
|
tms |
3 |
tm segs (best choice for TM segments) |
seq MBY pdb MBY lcl |
seq MBY pdb MBY lcl MBY tms |
|
tmf |
3 |
tm segs filtered |
seq MBY pdb MBY lcl |
seq MBY pdb MBY lcl MBY tmf |
|
lnk |
4 |
linkers |
seq MBY pdb MBY lcl MBY tms |
seq MBY pdb MBY lcl MBY tms MBY lnk |
|
lnk |
4 |
linkers |
seq |
seq MBY lnk |
|
ucd |
5 |
unchar domains |
seq |
seq MBY ucd |
|
cdo |
6 |
characterized domains |
seq |
seq MBY cdo |
|
alp |
7 |
alla segs |
seq MBY pdb MBY lcl MBY tms MBY lnk |
seq MBY pdb MBY lcl MBY tms MBY lnk MBY alp |
|
bet |
8 |
allb segs |
seq MBY pdb MBY lcl MBY tms MBY lnk MBY alp |
seq MBY pdb MBY lcl MBY tms MBY lnk MBY alp MBY bet |
|
nul |
9 |
full len segs |
seq |
seq MBY nul |
Disclaimer
All the matches reported by GeneCensus, particularly via the "find
folds in genomes" query box are based on simple automatic application
of sequence comparison program to the genomes -- in particular FASTA
with an e-value threshold of .01. As such, they are expected to
under-report the number of true-positive matches compared to more
elaborate approaches (though perhaps also have few false positives!).
Two specific limitations to the fold-usage analysis presented here are
discussed below.
Limitations of the Approach: The Small, Incomplete Number of Known
Folds
First, only a relatively small number of folds can be surveyed,
involving no more than a fifth of the ORFs in a genome. (This number
would be even smaller if one were to restrict attention to just the
ORFs in a genome that have been studied directly by
crystallography or NMR. For example, it is currently 52 out of 6218
for yeast, as reported by Sacc3D (Cherry et al., 1998).)
The situation is expected to improve in the future as new structures
are determined, but it will be a while before all the folds in a
genome are known -- especially considering that the increase in new
folds is much slower than the increase in new structures (Brenner
et al., 1997). An important corollary of this is that the absolute
counts found in a given genome survey are (usually) an
under-representation of the true numbers. Furthermore, they are
contingent on the evolving contents of the databank. Thus, over time
as more structures are added to the databank, one should expect such
statistics as the most common folds and number of shared folds to
change somewhat.
Comprehensive application of ab initio structure prediction and
advanced sequence-comparison and fold recognition methods to complete
genomes can overcome somewhat the limitations of only knowing a small
number of folds (Fischer & Eisenberg, 1997; Gerstein, 1997),
allowing one to survey the complete inventory of proteins in an
organism. However, in its present form, structure prediction is not a
substitute for structure determination, especially in situations where
the fold is completely new. Moreover (as discussed below) using
state-of-the-art sequence comparison methods introduces a measure of
variability and uncertainty into the results, as different methods
will give different results at the margin.
In summary, different comparison programs and fold databases will
give different numbers.
Limitations of the Approach: Biases in PDB and in the Genomes
In addition to rendering the results here, in a sense, incomplete, the
small number of known folds also means that the results may be
influenced to some degree by the biases in the PDB. These biases are
manifest in a number of ways.
First and most simply, there is a considerable disparity
between how often a fold occurs in the genomes (i.e. how many total
matches it has) and how often it occurs in the PDB (i.e. how many
known structures have this fold). This is indicated in Figure 1@@@
(and in the web presentation). One can immediately see how different
the common folds are in the PDB versus in the genomes. This
illustrates in a direct sense the biases in the PDB -- although these
sort of biases are not expected to affect the results (which are
principally concerned with "membership" rather than absolute
counts).
Second and more subtly, the composition of the PDB is biased
towards folds that occur in more heavily studied organisms such as EC
and SC. These biases are probably reflected in some of the results --
specifically, in the finding that there are many more known folds and
unique folds in the bacterium HI than in the archeon MJ, even though
both of these organisms have genomes of approximately the same
size.
Another subtle bias in the results here is in the selection of
genomes. The eight organisms picked were the first with complete
genomes to be sequenced, as has by necessity been done in all the
other multi-genome comparisons to date (e.g. Tatusov et al.,
1997). A more balanced comparison would perhaps have a more comparable
amount of eukaryotes and archaea to bacteria.